Breathing new life into drug delivery
Optimizing drug delivery through 3D airway modeling
We use anatomically accurate 3D models of the human upper airway to predict and control aerosol deposition–helping manufacturers develop safer, more reliable, and consistent drug products. We work with pharmaceutical companies, biotech startups, and generic manufacturers at any stage of respiratory drug development.
The challenge
Intersubject variability:
the greatest hurdle to respiratory
drug development
Bringing innovative respiratory drug products to market is challenging, costly, and filled with regulatory uncertainty.
Current in vitro methods often fail to predict in vivo performance because they can’t account for patients’ anatomical differences.
Clinical in vivo studies are expensive, complex, and can lack the sensitivity to detect critical formulation and device-related changes.
Our Solution
An in vitro testing platform that precisely captures intersubject variability

Full age-range coverage
We provide airway models for all key patient populations, including infants (3-24 months), pediatrics (2-11 years), and adults (21-75 years).

Representative Models
We analyzed deposition across 80 adult and pediatric nasal cavities to create low, mean, and high deposition model sets, accounting for population variability while minimizing costs.
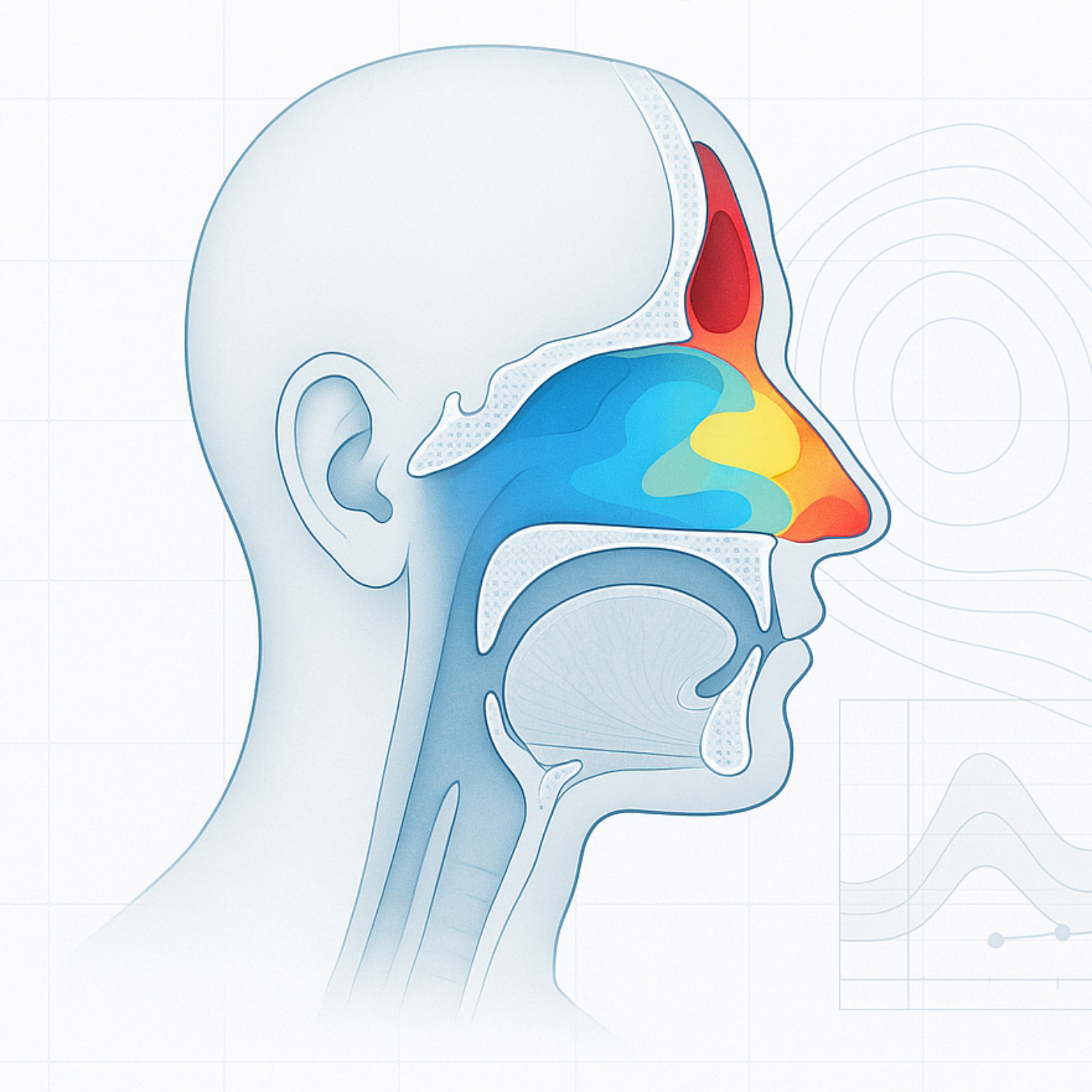
High-res regional data
Our models are segmented into precise anatomical regions, allowing exact identification of the product delivery area. This is unique to the 3DInhale platform.
Our platform
Specialized knowledge across key areas of respiratory drug development
From small molecules to biologics, our capabilities span formulation design to lifecycle management. We support reformulation, repositioning, and establishing bioequivalence of generics. And we can help you meet target product profiles aligned with therapeutic goals and regulatory expectations — while keeping patient experience and compliance at the heart of every solution.

Novel drug & device design
We support the design and evaluation of diverse nasal dosage forms, ensuring optimal delivery and patient usability. This includes metered-dose nasal sprays, powders, and aerosols that are gentle and user-friendly for sensitive populations.

Application areas
Our platform enables the development of respiratory drug products for both local and systemic applications, with a focus on non-invasive delivery.
We support noninvasive vaccines for mucosal immunity, direct nose-to-brain delivery for neurological pathologies (CNS targeting), and formulation strategies for potent, low-volume, targeted delivery of biologics.


Generics & Lifecycle management
Extend product value and ensure regulatory alignment through reformulation and bioequivalence strategies, including in vitro and in silico support for generic approval, as well as strategic updates to maintain your market relevance.
OuR Process
Support at any and every stage
We deliver a collaborative, data-driven suite of services
to support the full product development pipeline.
Technical
Design
We will hold an initial discovery meeting to understand your product goals and delivery strategy, offering feedback on optimized, patient-centric formulation, the device, and the delivery pathway. You will also receive guidance on regulatory alignment and defining the Quality Target Product Profile (QTPP).
Data Analysis
& Reporting
With our anatomically informed platform, we will conduct an in vitro deposition analysis, providing comprehensive formulation and device performance evaluations. These will be synthesized into custom reports with data visualizations and actionable recommendations.
Post-Development Support
Our team will take your product well beyond the finish line with lifecycle consulting for reformulation or a generic product strategy. We will also provide ongoing advisory support for your bioequivalence strategy, patient experience enhancements, and compliance.
3DInhale‘s anatomically accurate 3D airway models can optimize formulation targeting while accounting for intersubject variability. The separate sections of the nasal cavity can provide unique opportunities for drug delivery.
Anterior Nasal Segment
Supports the design of patient-friendly interfaces for intranasal devices, ensuring comfort and adherence while minimizing deposition variability.
Turbinate Regions
The superior turbinate enables CNS-targeted therapies through nose-to-brain delivery pathways. The middle and inferior turbinates are ideal for locally acting treatments (e.g., rhinitis, sinusitis) and systemic drug absorption due to their large surface area and vascularization.
Nasopharynx
Critical for noninvasive vaccine administration via the nose, leveraging its immunologically active region for effective mucosal immunization.
About us
3DInhale was founded at Virginia Commonwealth University (VCU) to address the critical, unmet need for patient-focused respiratory drug development.
Building upon the years of federally funded research, we strive to transform the development landscape with data-driven solutions that account for intersubject variability, reduce regulatory risk, and deliver safer, more effective products for patients who need them.

FOUNDER & CEO
Laleh Golshahi, Ph.D.
Laleh Golshahi, Ph.D., is the Founder and CEO of 3DInhale and the founding director of the Respiratory Aerosol Research and Education Laboratory at Virginia Commonwealth University. She holds three degrees in mechanical engineering including a Ph.D. Laleh joined VCU in 2012 and earned an Engineering Foundation Professorship in 2022. Dr. Golshahi’s work in respiratory drug delivery is globally recognized and she is the recipient of the 2019 Young Investigator by the International Society for Aerosols in Medicine. She is a recognized key opinion leader (KOL) in the field of respiratory drug delivery and has authored over 50 peer-reviewed publications and multiple book chapters on the subject. Based on more than five years of federally funded research at VCU, supported by the U.S. FDA, Dr. Golshahi has developed foundational knowledge and IP-protected innovations in intranasal drug delivery. Driven by a passion for creating respiratory drug solutions that work for everyone, regardless of age, gender, or dexterity, she founded 3DInhale to collaborate with key stakeholders across the nasal drug delivery ecosystem and make effective treatments accessible worldwide.
Partner with us.
Schedule a discovery meeting and learn how our platform can de-risk and optimize your product.
Acknowledgements
Thank you to the sponsors and mentors who have guided us along the way.






